Restriction Digest
Materials
- ddH20
- DNA sample on ice
- Appropriate 10X buffer (see below) on ice
- 10X BSA on ice (if necessary; see below)
- Restriction enzyme(s) (see below) on ice
- CIP (keep in freezer until needed)
Recipes
15 μL reaction
- 5 μL DNA
- 1.5 μL appropriate 10X buffer
- 1.5 μL 10X BSA (if needed—see below)
- Restriction enzyme(s) (see specific enzymes below for exact amounts)
- Don't immerse the tip, draw from the surface to avoid excess enzyme
- Add ddH20 to bring the final volume to 15 μL
Note: Use this recipe when verifying plasmid inserts
Note: You can check this reaction after 1 hour
30 μL reaction
- 12 μL DNA
- 3.0 μL appropriate 10X buffer
- 3.0 μL 10X BSA (if needed—see below)
- Restriction enzyme(s) (see specific enzymes below for exact amounts)
- Don't immerse the tip, draw from the surface to avoid excess enzyme
- Add ddH20 to bring the final volume to 30 μL
Note: Use this recipe when your DNA concentrations are sufficient
Note: For a BamHI–XbaI double-digest, use 3.3 μL of 10X buffer
40 μL reaction
- 20 μL DNA
- 4.0 μL appropriate 10X buffer
- 4.0 μL 10X BSA (if needed—see below)
- Restriction enzyme(s) (see specific enzymes below for exact amounts)
- Don't immerse the tip, draw from the surface to avoid excess enzyme
- Add ddH20 to bring the final volume to 40 μL
Note: Use this recipe when your DNA concentrations are low
Note: For a BamHI–XbaI double-digest, use 4.3 μL of 10X buffer
Procedure
- Combine ingredients for recipe
- Incubate at 37°C for 2.5 to 8 hours
- If preparing a ligation, add 0.5 μL CIP to the vector 30 minutes prior to ending the reaction
- When the reaction is complete you can store your samples in the -20°C freezer or proceed to in-gel ligation
Enzymes
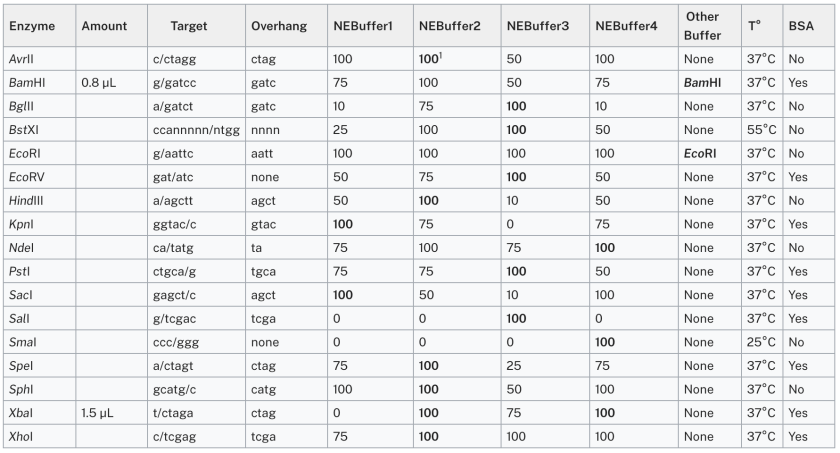
1Boldface indicates the preferred buffer for the enzyme.
Adapted from New England BioLabs 2005-06 Catalog and Technical Reference.
Double Digests
Suggested Buffers

1Sequential digest recommended.
2We actually prefer to do the double-digest and use Buffer 2 for the BamHI–XbaI double-digest.
Note that a double-digest of Bam HI and Xba I is not recommended. However, by following the procedure above you won't have any problems.
Adapted from New England BioLabs 2005-06 Catalog and Technical Reference.